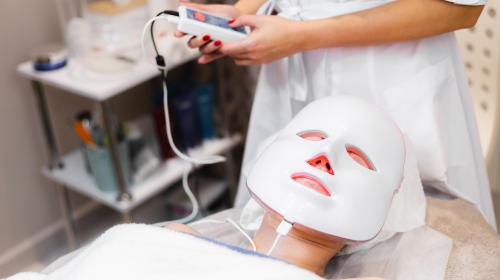
When does a wellness or beauty product become a medical device?
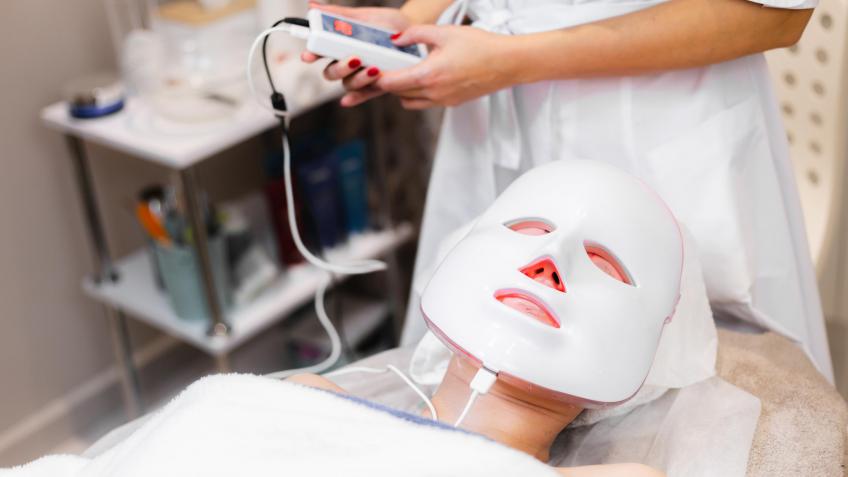
Recently, the UK’s medical regulator, the MHRA, has banned adverts for consumer focussed LED face masks for making claims such as ‘treating acne’ and ‘rosacea’ whilst not being registered as a medical device. Link to the BBC article here: https://www.bbc.co.uk/news/articles/cm2l8jldvjno.
At IDC, this is an issue of huge importance for our clients. With boundaries between health, wellness and beauty increasingly blurred, one question keeps coming up - ‘Will my new product be a medical device?’ This question matters for planning the new product development and for determining the cost and time of development. It also affects how the product will be marketed and sold. In reality, the answer is often not clear cut and, in many cases, product developers can have substantial leeway to determine whether or not to classify their ‘health’ product as a medical device. Here are six basic guidelines as you start to consider developing a health or beauty product:
1. Intent and Marketing Determine Classification
The single most important factor is intended purpose. Regulators in the UK, EU and US all classify a medical device based on what it is intended to do, not simply how it looks or operates. If a product is intended to diagnose, prevent, monitor, treat or alleviate disease or injury, it is considered a medical device. Conversely, if its purpose is purely cosmetic or to enhance wellbeing without a medical claim, it falls outside that scope. A wearable that “tracks heart rate for fitness” is a consumer wellness product, but if it “detects arrhythmias” or “alerts the user to potential heart conditions”, it’s medical. The intended medical purpose, whether explicit or implied through marketing materials, is decisive.
2. To Avoid Medical Regulation, Avoid Medical Claims
If you wish to claim that your product can diagnose, treat or prevent a disease or medical condition, then medical device regulations almost certainly apply. Likewise, you should be very cautious about claims of pain relief, circulation improvement, sleep disorders, or stress reduction associated with therapeutic outcomes.
3. Certain Products Will Always Be Medical Devices
Some products will always fall within the definition of a medical device and will therefore be subject to medical regulations. Any device which directly interacts with the body’s physiological functions. For example, anything that penetrates the skin, such as an injection pen, is implanted, or monitors a vital health parameter (e.g. a blood glucose) meter would generally always be regarded as a medical device.
4. US, UK or EU? Where You Sell Can Make a Difference
Some categories of products such as manual and electric toothbrushes, breast pumps and tampons are automatically classified as medical devices in the US but would only be classified as medical devices in UK or EU if the manufacturer wishes to make specific medical claims. Generally, the FDA is more likely to have blanket medical device classifications but check the rules for all markets that you are interested in.

5. Certifying as a Medical Device has Advantages as well as Costs
A medical device classification gives users confidence to trust the device and is a way to stand out in a crowded market. It also allows more specific claims to be made about the benefits and results of using the product. According to the MHRA, there are currently no LED light therapy masks that are approved as medical devices in the UK. Getting a Class IIa medical approval for an LED light mask that is proven to “treat acne” or “improve healing of scars” would offer a huge advantage over the competition.
6. A Non-Medical Product Still Needs to be Safe
If your company is importing and selling a consumer product in the market you are classified as the legal manufacturer (you need not be the company making the product) and therefore you are responsible for complying with all relevant laws, standards and regulations. The legal manufacturer should create and maintain a technical file demonstrating that the safety risks have been mitigated and regulatory requirements met. They should hold details of the technical specifications, including materials used and any safety tests carried out. Although a UKCA or CE mark can often be self-certified, you are legally required to hold this information.
IDC has wide experience of developing consumer wellness, beauty and medical products. If you have a potential product that you are looking to develop, we would be happy to discuss and advise on the development options, medical regulations, costs and timescales for developing your device.
About the Author
Stephen Knowles is the Managing Director of IDC. A chartered engineer with 25+ years experience leading technical and non-technical product developments in the medical industry. With a PhD in Mechanical Engineering and a thorough understanding of design and manufacture, Stephen is often invited to speak on the subject of product development. Contact: stephen.knowles@idc.uk.com

Sign up for our top resources on product design and development
Other articles you may be interested in