Medical Electronics: Bridging Innovation and Safety in Healthcare Devices
Creating electronics for medical devices is a unique design challenge, one where clinical safety, human usability, and technical performance must be expertly aligned. At Industrial Design Consultancy (IDC), we specialise in the development of high-quality medical electronics, delivering dependable solutions that function with precision in critical healthcare settings.
With decades of experience, in-house test facilities, and ISO 13485 certification, our multidisciplinary team transforms ideas into fully engineered systems ready for manufacture and regulatory approval.

Engineering Medical Devices for Real-World Use
Unlike consumer or industrial products, medical devices operate in highly variable environments, from busy operating theatres to unsupervised home care. Electronics within these products must be responsive, safe, and fail-safe by design.
At IDC, we develop embedded systems that are tightly integrated with the device’s mechanical and user-facing elements. Our team works across every aspect of electronics design: from schematic capture and PCB layout to firmware development and EMC testing. This integrated approach ensures that every function is aligned with how the device will be used by healthcare professionals or patients.
Our experience includes power management for portable devices, sensor integration, actuator control, and wireless communication, all tailored to the specific demands of regulated healthcare applications.
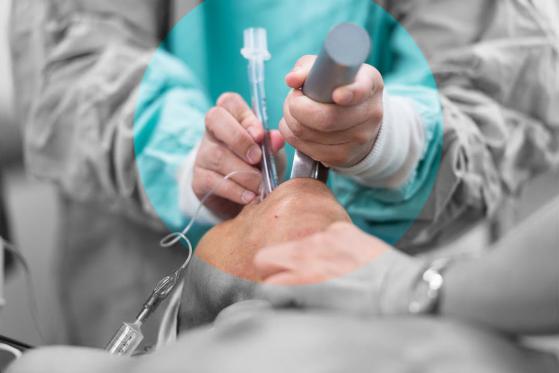
Compliance is more than a final checkbox, it is a design principle in every IDC project. Our engineers and designers work to internationally recognised medical standards from the beginning of development. Whether aligning to IEC 60601 for electrical safety or supporting FDA documentation requirements, we ensure that our designs are backed by robust, traceable processes. Informed by early clinical input and usability testing, we ensure that the electronics not only pass technical validation but also support safe and intuitive operation by end users. This balance of regulatory alignment and real-world practicality is central to our process. In our UK-based development centre, we carry out fast-paced design iterations supported by in-house testing. We simulate and prototype early to understand failure modes, measure system response, and optimise firmware logic under real-world constraints. This hands-on, test-driven process enables more confident engineering decisions and accelerates time-to-approval. Our facilities support rapid prototyping of both circuit boards and enclosures, and allow us to test across a wide range of parameters, from thermal performance to interaction accuracy.Designing with Regulatory and Clinical Insight
Test-Driven Development, In-House
Medical electronics must move efficiently from prototype to production without compromising quality or traceability. We engineer every system with manufacture in mind, applying rigorous DFM principles and working with suppliers to ensure that our designs are ready for scale-up. We also provide technical documentation packages, BOM management, and supplier handover support, giving clients a smooth path to volume production. Whether producing a small batch for clinical trials or preparing for global rollout, we help navigate the critical final steps.Designing for Manufacture, From the Ground Up
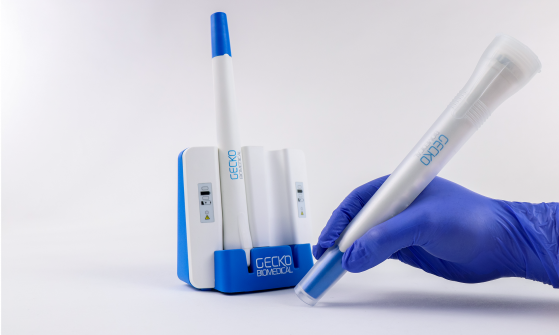
We developed the Setalum light source for Gecko Biomedical (now Tissium): a pen-sized, rechargeable 405nm curing device designed for use in complex vascular reconstructive surgery. With no off-the-shelf solution available, IDC's electronics, mechanical, and industrial design teams worked together to take the product from initial requirement to FDA-approved clinical trials in just 12 months. The electronics presented significant challenges in miniaturisation, with the entire system needing to fit within a pen-form factor. We used programmable logic to minimise component count and ensure a clear, compliant verification process under IEC 60601. A custom collimated lens and prism configuration was developed to maintain consistent beam intensity regardless of device orientation: a critical requirement identified through clinical observation and clinician interviews. Sterility was addressed through a disposable sterile sleeve combined with infrared sensors that prevent the light from activating unless the sleeve is correctly fitted. The reusable electronic device achieved Class I classification in the EU, while the sterile sleeve was classified as Class IIa - a carefully considered split that reflects the precision of our regulatory approach from the outset of development.Case Studies: Innovation in Action
Setalum Surgical Adhesive Curing Device - Gecko Biomedical (now Tissium)
For long-term partner XFT Medical, IDC engineered a pneumatic hand rehabilitation glove that blends EMG feedback with soft robotics to support stroke recovery. Designed for home use, the glove had to be simple to don with one hand, comfortable over long sessions, and intelligent enough to respond to patient signals. Our team handled everything from user research and ergonomic prototyping to system integration and manufacturability. Key design decisions included custom neoprene fabrication, integrated wrist support, and modular bellows for powered motion, all tuned to support the patient journey toward motor recovery.XFT Hand Rehabilitation Glove

From novel surgical tools to connected rehabilitation devices, our work in medical electronics consistently balances safety, innovation, and performance. Whether you're developing a diagnostic device, wearable therapy system, or next-generation instrument, IDC is equipped to take your idea through engineering, compliance, and production. Visit our medical electronics page to see more of what we do, or get in touch to begin your next development journey.Partnering for Success in Medical Electronics