XFT
Foot Drop Device
Helping stroke victims
to walk normally
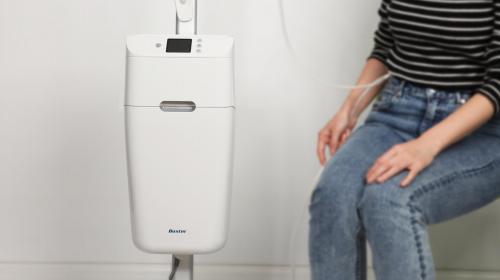
IDC developed the first all-in-one footdrop device, with fully integrated electrodes, to aid neurological walking problems.
IDC worked with XFT to develop the fourth generation of its foot drop device which helps patients with certain neurological conditions walk again. The device marks a great advance in technology, being the first device of its kind to have fully integrated electrodes moulded right into the strap in an all-in-one design.
Having already completed the second and third generations of the foot drop device, with the previous version being the thinnest wearable device available for foot drop treatment, IDC was delighted to once again push the boundaries of wearable medical technology.
The foot drop device works by delivering electrical pulses to counteract the effect of the dropping of the foot due to nerve damage or leg muscle paralysis. It works by stimulating the fibula and anterior tibia nerves so muscles raise the foot at the appropriate time to help train the patient to restore neurological motor function. This works in combination with an integrated gyroscopic sensor, which detects the walking rhythm and helps regulate a normal walk.
XFT came to IDC with the idea to develop a fully integrated device with built-in electrodes, and needed IDC’s expertise to develop a solution that would work universally on either left or right legs. Unlike previous generations of the product, the G4 was designed to be used in a hospital as a multi-patient device. This meant that it needed to be waterproof, easy to clean and adaptable to all sizes and shapes of legs.
IDC’s team initially spent time researching products and users to gain insight into the needs of users. The team also worked with medical staff in hospitals to understand all elements of product use and medical requirements. IDC’s industrial designers and engineers then used this insight to design a solution.
With wide variation in the size of people’s legs and calf profiles, it was challenging to develop an electrode solution that took account of different nerve positions. Electrode development was therefore a key focus of the project. The team worked with XFT to gather test data about the effectiveness of various electrode positions on different shaped legs. This data was then used to map nerve areas so a universal electrode shape could be defined.
Once a concept was developed, it was tested and refined using rapid prototyping, to ensure the optimum shape and position of the electrodes.
Another key element of the development was wearability and comfort, and the team had to find the best materials and structural solution, as well as ensuring easy cleaning for medical staff.
After researching materials, TPE was chosen for the device, but the stainless steel electrodes would not bond easily with TPE, so an innovative structure was developed with a semi-flexible PP substrate. This allowed the electrodes to flex and maintain electrical contact while integrated into the TPE. The manufacturing process required to achieve this was quite complex, with stainless steel pressings over-moulded into a 6-electrode semi-flexible array, which was then over-moulded by a unibody elastomer strap.
A user-interface was incorporated into the device which was designed to automatically flip 180° according to which leg the user was wearing it on. User friendliness was also enhanced by a simple solution to tighten the strap single-handedly.
As the third XFT footdrop device to be designed by IDC, the team once again broke boundaries in delivering the market's first all-in-one foot-drop device with fully integrated electrodes.